About our Lab
Viral Immunology
The main focus of our labortory's research is studying how two unrelated respiratory viruses affect each other’s ability to infect and replicate within cells, and cause disease in the host. We use cell culture models of respiratory epithelial cells and infection in mice to address this.
Current Research
Funding Agency - National Institute of Health
Currently, our lab is working to identify the relationships between the biological variables of respiratory viral co-infection and the resulting disease severity. These variables include viral stains, doses, viral timing, genetic variation in the host, immune response (which is critical for viral clearance), and pathology. In order to understand how these complex interactions dictate co-infection outcome, we are establishing a murine model of respiratory viral co-infection and use it to control, quantify, and model how viruses interact within the respiratory tract at the viral, cellular, and organismal level within the contect of the murine host's immune system. Infection of human and murine respiraory viruses in respiratory epithelial cells, perfomed in paralell with our other experiments, will help determine the generality of our model to human infections.
These experiments will allow us to test our "central hypothesis" that respiratory co-infections change disease severity compared to single infections both by direct viral interactions and by modulating host responses, resulting in altered clearance of the individual viruses and/or immune-mediated pathology. To test our hypothesis we will rigorously test them through two specific aims.
- Aim 1 - Determine how Viral co-infection variable influence viral load, host responses, pathology, and disease severity in mice
- Aim 2 - Determine the effects of co-infections on viral growth dynamics and cellular responses in respiratory epithelical cells in vitro
Experimental Setup
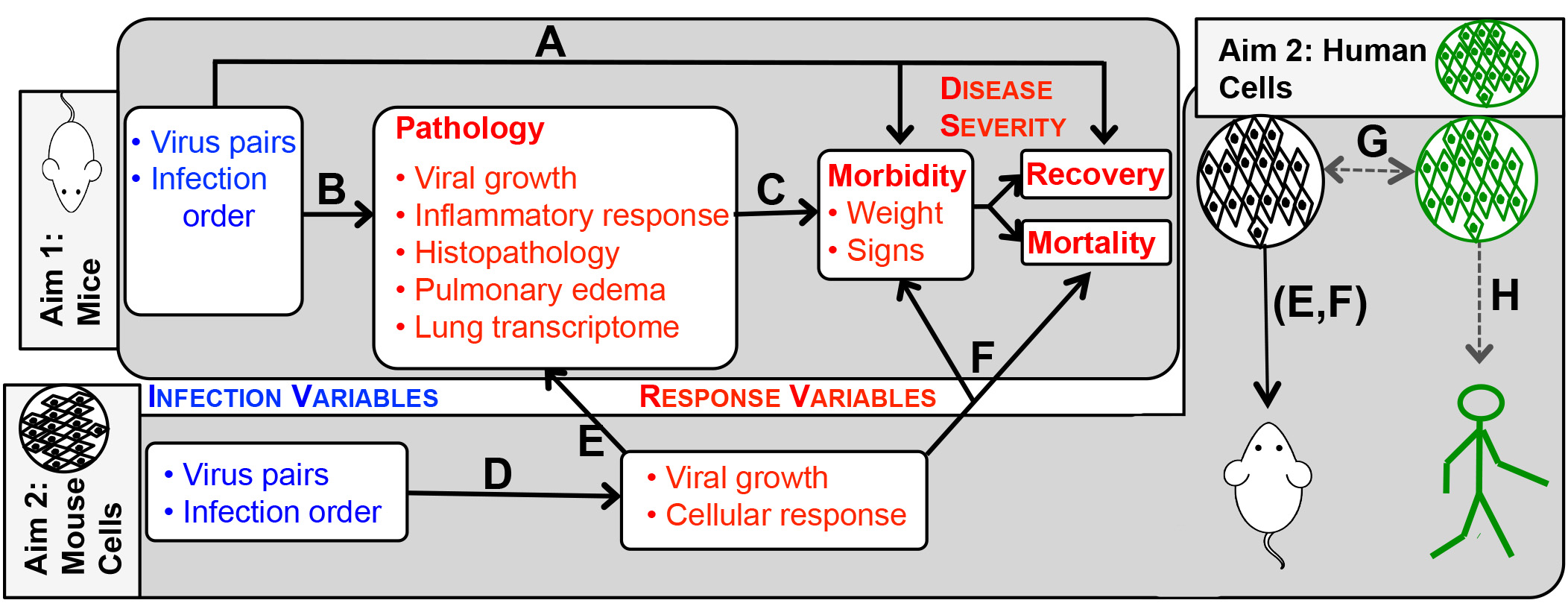
The figure above shows the process of modeling experimental data in aims 1 and 2. Letters indicate processes we will model based on experimental data. A-D are within-aim analyses, E and F bridge the two aims, G will determine the generality of our murine data to human cells and viruses, and H will use these findings to predict outcomes of human respiratory co-infections. Aim 1 will analyze: (A) the role of co-infection variables (blue) in determining disease severity (morbidity and mortality), (B) how different co-infection variables alter pathology response variables and (C) how well pathology explains disease outcomes. In Aim 2, we will determine (D) how the different co-infection variables affect in vitro response variables (viral growth and cellular gene expression). We will then determine how responses to co-infection in vitro can predict (E) pathology and (F) disease responses in vivo. The portion of Aim 2 on the right hand side shows how modeling data from murine and human cell systems (G) will be combined with cell-to-animal modeling (E,F) to make predictions about co-infection in humans (H).
Research Team
University of Idaho - Biological Sciences - Gibb Hall 107

Dr. Tanya Miura
Associate Professor - Principal Investigator
Ph.D. Microbiology, Colorado State University, 2000
B.A. Zoology, Honors Program, The University of Montana, 1995
Postdocoral Fellowship - National Jewish Hospital - Denver, Colorado
Postdocoral Fellowship - University of Colorado Health Sciences Center - Denver, Colorado
Contact: tmiura@uidaho.edu

Andy Gonzalez
PhD Graduate Student - Viral Immunology
B.S. Animal Sciences, University of Idaho, 2013
M.S. Microbiology - Immunology, Pathology Colorado State University, 2014

Joseph DeAguero
Ph.D. Graduate Student - Biomathmatics
Bioinformatics and Computational Biology Fellow
B.S. Applied Mathematics - University of New Mexico, 2013

Elijah Benson
Undergraduate Research Assistant
Idaho IDeA Network Biomedical Research Excellance - INBRE
B.S. Microbiology - University of Idaho, 2018

Emmanuel Ijezie
Undergraduate Research Assistant
B.S. Microbiology - University of Idaho, 2018

Frankie Scholz
Undergraduate Research Assistant
B.S. Microbiology - University of Idaho, 2019

Bhim Thapa
Laboratory Manager - Research Technician
B.S. Biology - Washington State University
Contact: bhim@uidaho.edu; (208) 885-6044
Past Lab Members

Jade Rodgers
Undergraduate Research Assistant
Idaho IDeA Network Biomedical Research Excellance
B.S. Biology, University of Idaho, 2015

Audrey Harris
Masters Graduate Student
B.S. Biology, Washington State University, 2010

Alicia Healey
Undergraduate Research Assistant
Research Experience for Undergraduates - REU
B.S. Biochemistry - Simmons College, 2018

Anna Rodriguez
Research Technician
B.S. Animal Science - University of Idaho, 2016

John Clary
Laboratory Manager - Research Technician
B.S. Biology - Walla Walla University, 2013
M.S. Cancer Immunology - Walla Walla University, 2016
Recent Lab News
“Co-Infection with Rhinovirus and Influenza A virus in vitro”

“The Old Ball and Chain: Effects of Tetherin on Diverse Respiratory Viruses”

“Target Cell Limitation Model Derived with in Vitro Data to Understand Viral Co-infection”

“Can the common cold protect you from influenza? A murine model of respiratory viral coinfection”
“Target Cell Limitation Model Derived with in Vitro Data to Understand Viral Co-infection”

Oct. 16th, 2015 - Andy Gonzalez wins first place at 2015 IDeA conference in Couer D'Alene
"Can the common cold protect you from influenza? A murine model of respiratory viral coinfection"

Colorado State Univeristy - Can the common cold protect you from influenza? A murine model of respiratory viral coinfection

Awards and Grants
Awards
Dr. Tanya Miura wins Mid Career Presidental Award
Biology faculty member, Dr. Tanya Miura, is the recipient of the 2016 Presidential Mid-Career Award. This award was established in 2011-12 to acknowledge faculty, usually during the early to middle stage of their career, who have demonstrated a commitment to outstanding scholarship, teaching and engagement. Recipients of this award will be considered as some of the University’s most gifted faculty members who continually serve as role models, a source of inspiration for students, and whose scholarship contributes to the intellectual development and lives of people in Idaho and globally.
-
Anna Rodriguez wins NAIPI-VSA-Sponsored ‘Take Your IDeA Science to Capitol Hill’
Anna Rodriguez was nominated and rewarded the NAIPI (National Association of IDeA) for her out standing research in A Target Cell Limitation Model Derived iwth in vitro Data to Understand Viral Co-Infection.
-
Elijah Benson wins College of Science Hill Fellowship
“Bound and gagged: effects of tetherin against diverse respiratory viruses”, Tanya Miura, mentor
Grants
-
Disease severity during viral co-infection - University of Idaho
-
New Perspectives on Ebola Virus Evolution.
-
Initiating a watch list for Ebola virus antibody escape mutations.
-
Proteomic analysis reveals down-regulation of surfactant protein B in murine type II pneumocytes infected with influenza A virus.
-
Neutrophils are needed for an effective immune response against pulmonary rat coronavirus infection, but also contribute to pathology.
-
Differentiated phenotypes of primary murine alveolar epithelial cells and their susceptibility to infection by respiratory viruses.
-
Virus-infected alveolar epithelial cells direct neutrophil chemotaxis and inhibit their apoptosis.
-
Rat respiratory coronavirus infection: replication in airway and alveolar epithelial cells and the innate immune response.
-
Host-pathogen interactions during coronavirus infection of primary alveolar epithelial cells.
-
E1A oncogene enhancement of caspase-2-mediated mitochondrial injury sensitizes cells to macrophage nitric oxide-induced apoptosis.
-
SARS-CoV replicates in primary human alveolar type II cell cultures but not in type I-like cells.
-
The spike glycoprotein of murine coronavirus MHV-JHM mediates receptor-independent infection and spread in the central nervous systems of Ceacam1a-/- Mice.
-
Rat coronaviruses infect rat alveolar type I epithelial cells and induce expression of CXC chemokines.
-
The interaction of adenovirus E1A with p300 family members modulates cellular gene expression to reduce tumorigenicity.
-
Rat coronavirus infection of primary rat alveolar epithelial cells.
-
Expression of an E1A/E7 chimeric protein sensitizes tumor cells to killing by activated macrophages but not NK cells.
-
E1A oncogene-induced sensitization of human tumor cells to innate immune defenses and chemotherapy-induced apoptosis in vitro and in vivo.
-
Adenovirus E1A, not human papillomavirus E7, sensitizes tumor cells to lysis by macrophages through nitric oxide- and TNF-alpha-dependent mechanisms despite up-regulation of 70-kDa heat shock protein.
IDeA Network of Biomedical Research Excellence (INBRE)
Summer Research Experiences for Undergraduates (REU)
If you are interested in undergraduate research in the Miura laboratory sponsered by INBRE or REU, please click on the provided links and follow the application process. For more information or questions please contact tmiura@uidaho.edu
National Institue of Health - National Institute of General Medical Sciences
Project # - 1P20GM104420-01A1
Publications
January 2003 - August 2016
Brown CJ, Quates CJ, Mirabzadeh CA, et al. New Perspectives on Ebola Virus Evolution. PLoS ONE. 2016;11(8):e0160410.
Miller CR, Johnson EL, Burke AZ, et al. Initiating a watch list for Ebola virus antibody escape mutations. PeerJ. 2016;4:e1674.
Kebaabetswe LP, Haick AK, Gritsenko MA, et al. Proteomic analysis reveals down-regulation of surfactant protein B in murine type II pneumocytes infected with influenza A virus. Virology. 2015;483:96-107.
Haick AK, Rzepka JP, Brandon E, Balemba OB, Miura TA. Neutrophils are needed for an effective immune response against pulmonary rat coronavirus infection, but also contribute to pathology. J Gen Virol. 2014;95(Pt 3):578-90.
Kebaabetswe LP, Haick AK, Miura TA. Differentiated phenotypes of primary murine alveolar epithelial cells and their susceptibility to infection by respiratory viruses. Virus Res. 2013;175(2):110-9.
Rzepka JP, Haick AK, Miura TA. Virus-infected alveolar epithelial cells direct neutrophil chemotaxis and inhibit their apoptosis. Am J Respir Cell Mol Biol. 2012;46(6):833-41.
Funk CJ, Manzer R, Miura TA, et al. Rat respiratory coronavirus infection: replication in airway and alveolar epithelial cells and the innate immune response. J Gen Virol. 2009;90(Pt 12):2956-64.
Miura TA, Holmes KV. Host-pathogen interactions during coronavirus infection of primary alveolar epithelial cells. J Leukoc Biol. 2009;86(5):1145-51.
Radke JR, Siddiqui ZK, Miura TA, Routes JM, Cook JL. E1A oncogene enhancement of caspase-2-mediated mitochondrial injury sensitizes cells to macrophage nitric oxide-induced apoptosis. J Immunol. 2008;180(12):8272-9.
Mossel EC, Wang J, Jeffers S, et al. SARS-CoV replicates in primary human alveolar type II cell cultures but not in type I-like cells. Virology. 2008;372(1):127-35.
Miura TA, Travanty EA, Oko L, et al. The spike glycoprotein of murine coronavirus MHV-JHM mediates receptor-independent infection and spread in the central nervous systems of Ceacam1a-/- Mice. J Virol. 2008;82(2):755-63.
Miura TA, Wang J, Holmes KV, Mason RJ. Rat coronaviruses infect rat alveolar type I epithelial cells and induce expression of CXC chemokines. Virology. 2007;369(2):288-98.
Miura TA, Cook JL, Potter TA, Ryan S, Routes JM. The interaction of adenovirus E1A with p300 family members modulates cellular gene expression to reduce tumorigenicity. J Cell Biochem. 2007;100(4):929-40.
Miura TA, Wang J, Mason RJ, Holmes KV. Rat coronavirus infection of primary rat alveolar epithelial cells. Adv Exp Med Biol. 2006;581:351-6.
Miura TA, Li H, Morris K, et al. Expression of an E1A/E7 chimeric protein sensitizes tumor cells to killing by activated macrophages but not NK cells. J Virol. 2004;78(9):4646-54.
Cook JL, Miura TA, Iklé DN, Lewis AM, Routes JM. E1A oncogene-induced sensitization of human tumor cells to innate immune defenses and chemotherapy-induced apoptosis in vitro and in vivo. Cancer Res. 2003;63(12):3435-43.
Miura TA, Morris K, Ryan S, Cook JL, Routes JM. Adenovirus E1A, not human papillomavirus E7, sensitizes tumor cells to lysis by macrophages through nitric oxide- and TNF-alpha-dependent mechanisms despite up-regulation of 70-kDa heat shock protein. J Immunol. 2003;170(8):4119-26.
Research Opportunities
University of Idaho - Biological Sciences
At the University of Idaho, there is an abundance of opportunities that undergraduates and graduates can participate in cutting edge research. For undergraduates, engaging in research enhances their abilities to succeed as young scientist. At the University of Idaho there are two specific programs dedicated to helping undergraduates partake in research:

