Research
Local Adaptation
Coevolution
Transmissible Vaccines

Local Adaptation
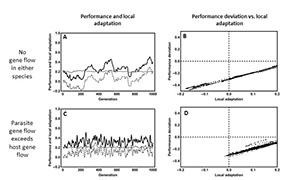
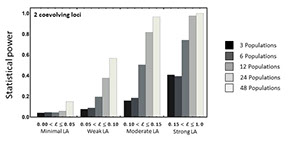
Understanding the causes and consequences of local adaptation has long been a focus of research in the lab. Questions we continue to pursue include: 1) Why do some species evolve strong patterns of local adaptation whereas others do not? 2) Why do some parasites perform better on sympatric host populations whereas others perform better on allopatric host populations? 3) What do studies of local adaptation tell us about the genetics of species interactions?
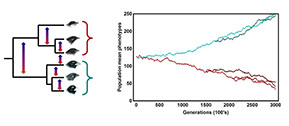
Coevolution
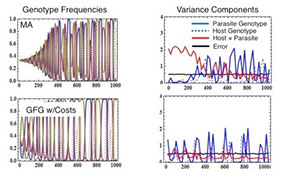
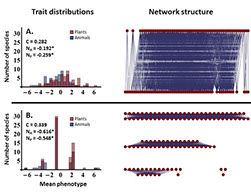
The majority of work in the lab revolves in some way around the potential for interacting species to coevolve. The coevolutionary questions we are currently focused on include: 1) Can we develop statistical methodologies that allow the strength of coevolution to be estimated from phenotypic data? 2) Is it possible to identify the genes mediating coevolutionary interactions in locally adapted systems? 3) What role does coevolution play in community structure and function?, and 4) What practical insights can be gained through the perspective of coevolutionary medicine?
More information:
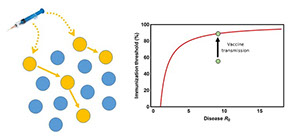

Transmissible Vaccines
The development of viral vaccines has had remarkable and long-lasting impacts on human health and on the health of domestic and wild animal populations. Despite these impressive successes, many infectious diseases cannot yet be efficiently controlled or eradicated through vaccination programs because it is impossible to vaccinate a sufficient proportion of the population to guarantee herd immunity. This problem is particularly acute for diseases of wildlife, including emerging infectious diseases such as Ebola, MERS, and Rabies where direct vaccination is impractical, cost-prohibitive, or even impossible.
Recent advances in molecular biology suggest that the centuries-old method of individual-based vaccine delivery could be on the cusp of a major revolution. Specifically, genetic engineering brings to life the possibility of a transmissible or “self-disseminating” vaccine. Rather than directly vaccinating every individual within a population, a transmissible vaccine would allow large swaths of the population to be vaccinated effortlessly by releasing an infectious agent genetically engineered to be benign yet infectious. This concept may sound like science fiction, but the oral polio vaccine already does this to a limited extent, and transmissible vaccines have now been developed and deployed in wild animal populations. We are working to develop a mathematical theory for predicting when such transmissible or self-disseminating vaccines will be epidemiologically effective and evolutionarily robust.
More information on our work with transmissible vaccines can be found through the links below:
Evaluating the Promise of a Transmissible Vaccine